PAI
Moderated by Prof. Dr. Gijs van Soest, professor in Invasive Imaging, Thoraxcentre BME group, Erasmus MC, Rotterdam, The Netherlands
Photo-Acoustic Imaging (PAI) is an optical imaging techniques which relies on different absorption spectra of (de-)oxygenated and total hemoglobin (HbO, HbR and HbT). Through the mechanism of Neurovascular Coupling (NVC), these measurements allow for an indirect detection of functional activation (1-3).
Technical Parameters
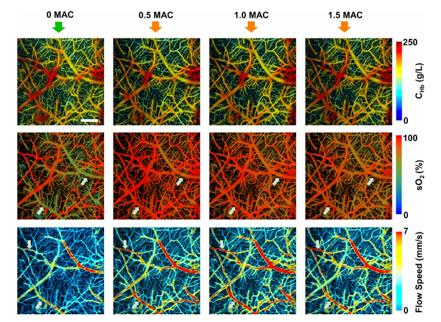
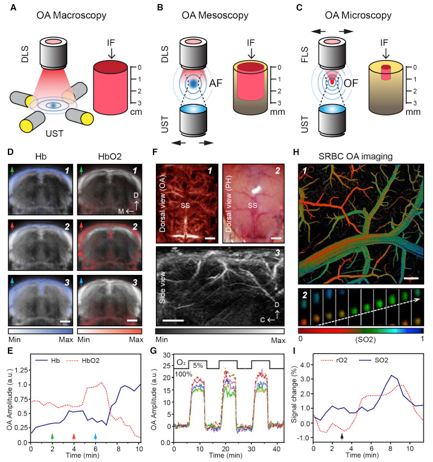
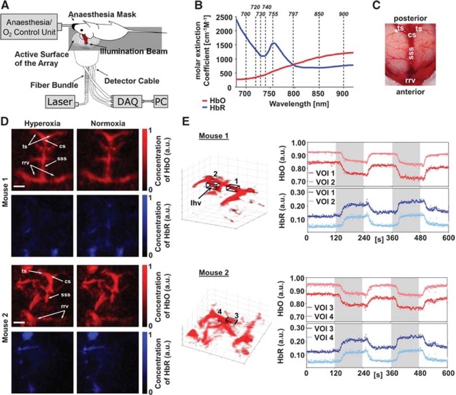
In PAI, optical excitation is combined with acoustic detection (1-9). After illuminating the tissue with a laser pulse, chromophores such as RBCs absorb the optical energy depending on the oxygenation status of their hemoglobin and the wavelength, which leads to a transient, localized rise in temperature. (4-7). Due to thermoelastic expansion, the absorbing RBCs generate an acoustic wave, which can be picked up by an ultrasound transducer. Because PAI can combine the high-contrast benefits of optical imaging, with potential for chemical specificity, while bypassing the optical diffusion limits on penetration by detecting ultrasound instead, it can reach significant penetrative depths, in the range of centimeters. Here again, there is a scalable trade-off between spatial resolution and depth penetration, depending on the ultrasonic center frequency used. In fPAM, one aims primarily for high spatial resolution (<50 µm) using a raster scanning method, with as a consequence relatively low penetration depths (millimeters) (8). In functional Photoacoustic Tomography (fPAT), also known as Photoacoustic Computed Tomography (PACT), the acoustic waves emitted from the tissue can be detected by an array of ultrasonic transducers at multiple angles simultaneously, allowing for cross-sectional or volumetric imaging (8,10,11). Here, penetrative depths of several centimeters have been described (2). So far, the literature reports cases of 5D-fPAT, which involves real-time, multispectral visualization, allowing for volumetric detection of multiple spectrally distinctive – and functionally informative – hemodynamic changes in the brain (11).
Biological Substrate
Using multispectral PAI and exploiting the fact that different functionally informative absorbers are sensitive to specific wavelengths, functional neuroimaging can be achieved. As displayed in Figures 1-3, distributions of e.g. HbO en HbR can be retrieved by spectral processing of images using discrete wavelengths. From there, other parameters such a HbT (HbO + HbR), SO2 (HbO/(HbO+HbR)) and CBV (the number of voxels for which the HbT signal is higher than a given threshold) can be determined (7,9,11). As discussed previously in the chapters on fNIRS, fUS and OCT, being able to detect these metabolic and/or hemodynamic-related changes can be an indirect measure to detect functional neuronal activation.
Intra-operative applicability
Compared to other optical techniques, PAI has the strong advantage of being able to combine the contrast of optical imaging, the relative depth penetration of ultrasound, while remaining contrast-free. Much has been written about the application of PAI for structural as well as functional brain imaging in a pre-clinical setting (7,8), however, actual intra-operative applications have not been described as of yet (12). The main bottleneck for this technique in the intra-operative setting will most likely be the still limited penetrative depth, which will allow for superficial cortical imaging only (12).
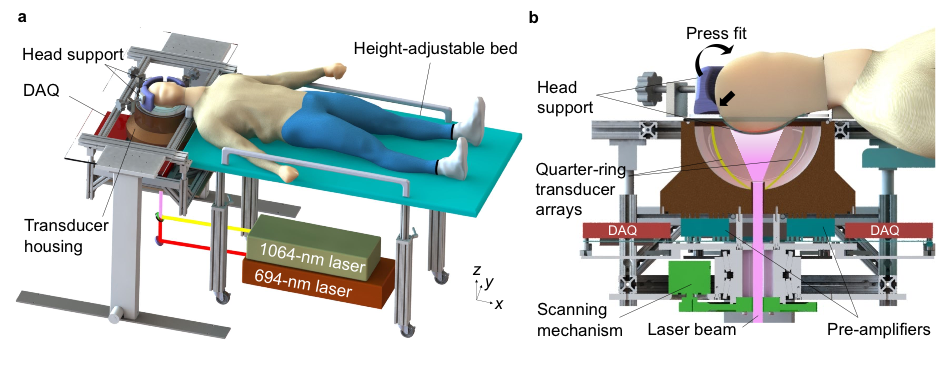
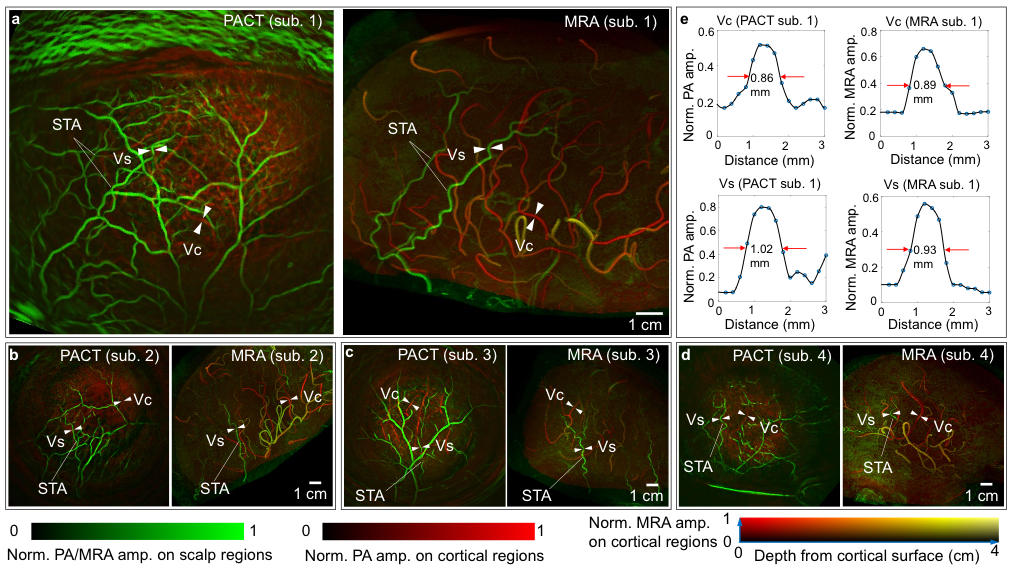
Recently, the first successful in-human brain imaging application was described in literature using a new 3D fPACT system, validated to fMRI (13,14). In this study, Na et al. (13) demonstrate the 1k3D-fPACT system which uses >1000 parallel ultrasonic transducer elements which are evenly distributed on a hemispherical bowl (Figure 4). In their application on hemicraniectomy patients, the study group was able to produce tomographic images of the brain with a wide field of view (10 cm), high spatial resolution (<500 µm) and reasonable penetration depth of around 1 cm. Both their angiography images (Figure 5) as well as their functional cortical maps during language and motor tasks were compared to similar images as produced in a 7T MRI system. The authors report how the technique was able ‘to detect functional activation faster than BOLD fMRI’ with ‘potentially greater specificity’ (13).
References
- Lin L, Hu P, Tong X, et al. High-speed three-dimensional photoacoustic computed tomography for preclinical research and clinical translation. Nat Commun. 2021
- Yao J, Wang L V. Photoacoustic tomography: Fundamentals, advances and prospects. Contrast Media and Molecular Imaging. 2011.
- Wang L V., Hu S. Photoacoustic tomography: In vivo imaging from organelles to organs. Science. 2012
- Li L, Zhu L, Ma C, Lin L, Yao J, Wang L, et al. Single-impulse panoramic photoacoustic computed tomography of small-animal whole-body dynamics at high spatiotemporal resolution. Nat Biomed Eng. 2017
- Wang L V., Hu S. Photoacoustic tomography: In vivo imaging from organelles to organs. Science. 2012
- Zhang P, Li L, Lin L, Shi J, Wang L V. In vivo superresolution photoacoustic computed tomography by localization of single dyed droplets. Light Sci Appl. 2019
- Cao R, Li J, Ning B, Sun N, Wang T, Zuo Z, et al. Functional and oxygen-metabolic photoacoustic microscopy of the awake mouse brain. 2017;
- Wang L V., Gao L. Photoacoustic microscopy and computed tomography: From bench to bedside. Annual Review of Biomedical Engineering. 2014
- Ovsepian S V., Olefir I, Westmeyer G, Razansky D, Ntziachristos V. Pushing the Boundaries of Neuroimaging with Optoacoustics. 2017.
- Tang J, Coleman JE, Dai X, Jiang H. Wearable 3-D Photoacoustic Tomography for Functional Brain Imaging in Behaving Rats. Sci Rep. 2016
- Gottschalk S, Felix Fehm T, Luís Deán-Ben X, Razansky D. Noninvasive real-time visualization of multiple cerebral hemodynamic parameters in whole mouse brains using five-dimensional optoacoustic tomography. J Cereb Blood Flow Metab. 2015
- Steinberg I, Huland DM, Vermesh O, Frostig HE, Tummers WS, Gambhir SS. Photoacoustic clinical imaging. Photoacoustics. 2019
- Na S, Russin JJ, Lin L, et al. Massively parallel functional photoacoustic computed tomography of the human brain. Nat Biomed Eng. May 2021
- Na S, Russin J, Lin L, et al. Mapping human brain function with massively parallel high speed three-dimensional photoacoustic computed tomography. SPIE-Intl Soc Optical Eng. 2021
